How to avoid issues a Medical Biller might face while bundling codes for Laboratory tests and panels?
Clinical Laboratories deal with a variety of different sections of clinical diagnosis categories from serological to histopathological and more. Billing these codes seems straight forward until the various laboratory start mixing up and elevate the chances of their denial.
In this article we discuss about bundling of panel codes and how we get through their billing fairly easily
What is ‘Panel’?
Packaging a series of akin lab tests into one comprehensive lab test is called as a panel or lab profile. That’s when the individual tests are bundled in such a way that they collectively help in clinical diagnosis and treatment of the patient.
Three terms that billers need to be familiar with, when dealing with Laboratory codes:
- Comprehensive Code – that defines a panel of bundled set of tests and their codes
- Component Codes – that defines each individual test that is part of that comprehensive code.
- Custom Panels – that defines a set of tests in a panel that are customized by the laboratory to their liking
Components of Lab test panels or profiles can be billed separately however CMS has also designed HCPCs code for each panel. So, they can be billed as bundled. Keeping in mind each lab test panel share a unanimous goal of identifying a singular prognosis in mind
Some Common Chemistry Panels are as follows:
- Basic Metabolic Panel (BMP) – tests that measure blood sugar (glucose) levels, electrolyte and fluid balance, and provide information about kidney function, respiratory function and liver function.
- Comprehensive Metabolic Panel (CMP) – provides the same information as the BMP as well as the status of the liver and key blood proteins.
- Electrolyte Panel – for detecting a problem with the body’s fluid and electrolyte balance.
- Lipid Profile – to assess the risk of developing cardiovascular disease.
- Hepatic Function Panel or Liver Panel – to screen for, detect, evaluate, and monitor acute and chronic liver inflammation (hepatitis), liver disease and/or damage.
- Renal Panel or Kidney Function Panel – includes tests to evaluate kidney function, such as albumin, creatinine, BUN, eGFR.
- Thyroid Function Panel – to assess thyroid gland function and help diagnose thyroid disorders.
For example
When a physician orders “Basic metabolic panel” or “comprehensive metabolic panel”, they actual mean the beneficiary has to get a series of (component) tests done in order to form a clear picture of your current health status and issues, if any.
Some panel codes can be a part of other more comprehensive panels as well, for instance a basic metabolic panel is a part of comprehensive metabolic panel. Meaning, its tests are already bundled into the comprehensive panel. If you are billing a Comprehensive Metabolic panel, then Basic Panel might not be reimbursed or any of its duplicate component codes billed separately.
What are guidelines for billing panel codes?
CMS is adamant and very clear about their policies regarding unbundling Laboratory panel codes, let’s have a look at its policy:
- Individual component lab tests are not to be repeated or duplicated.
- CMS will not pay for the panel code unless all of the tests in the definition are performed.
- If the laboratory has a custom panel that includes other tests, in addition to those in the defined CPT or HCPCS panels, the additional tests, are billed separately in addition to the CPT or HCPCS panel code.
- If a laboratory chooses, it can bill each of the component tests of these panels individually, but payment will be based upon the above rules.
- Laboratories shall bill the HCPCS panel test code and not unbundle the individual components if ALL components of the HCPCS panel are performed.
- If a group of tests overlaps two or more panels, the panel that incorporates the greater number of tests to fulfill the code definition should be reported and the remaining tests should be reported using individual test codes.
- Claims will be returned as processable/rejected if the HCPCS panel test code is not billed.
- Payment for the total panel may not exceed the sum total of the fee amounts for individual covered tests
This billing policy applies when:
a). Submitting a complete organ disease panel; or
b). Submitting individual component tests of an organ disease panel when all components of the panel were not performed
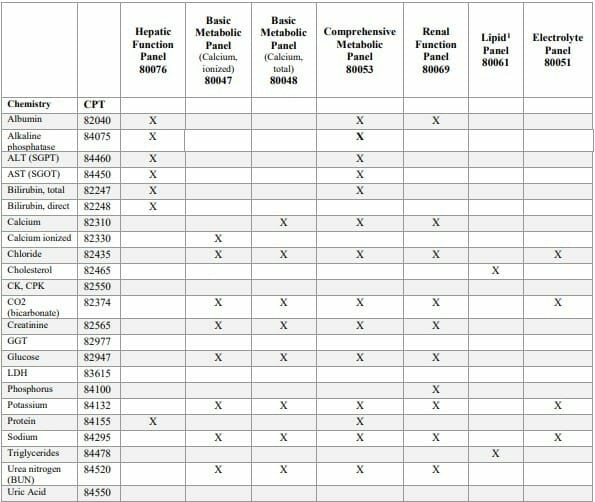
Following is a table released by CMS, which can be helpful in figuring out which component codes are bundled into the seven primary comprehensive panels.
Tests Performed More Than Once on the Same Day?
However, there will be certain instances where the ordering Provider might feel the requirement that some tests should be repeated for medically necessary reasons. Below we will touch how to deal with the bundling of panel codes and billing repeated and similar lab tests on a same day.
In that case, we have two Modifiers that can be appended to let the payers know why the lab tests were repeated and they were indeed medically necessary to perform again.
Modifier 91 Repeated clinical diagnostic laboratory test is used to report laboratory tests performed more than once on the same date to obtain subsequent, multiple test results. This modifier is not to be used when test is repeated due to specimen mishandling, insufficient sampling or re-confirmation.
Modifier 59 describes a distinct procedural service, and is used to identify procedures and services that are not normally reported together.
Let’s have a look how CMS guides us about their use:
Proper Use | Improper Use |
|---|---|
These modifiers may be used to indicate that a test was performed more than once on the same day for the same patient, only when it is necessary to obtain multiple results in the course of treatment. | These modifiers may not be used when tests are rerun to confirm initial results; due to testing problems with specimens and equipment; or for any other reason when a normal, one-time, reportable result is all that is required. |
It is important to understand that the 91 modifier is used for repeated laboratory tests specifically and Modifier 59 can be used to distinct two similar lab tests. | These modifiers may not be used when there are standard HCPCS codes available that describe the series of results (e.g., glucose tolerance tests, evocative/suppression testing, etc.). |
These modifiers may be used only for laboratory tests paid under the clinical laboratory fee schedule. |
Final say?
When it is necessary to obtain multiple results in the course of treatment, the modifiers 59 or 91 are used to indicate that a test was performed more than once on the same day for the same patient Improper use of modifiers is likely to indicate a fraudulent or abusive circumstance. These modifiers have very narrow application and that any evidence of excessive use will be referred for further review.
It would also be greatly helpful if Laboratories document the verbal communication from physician or other qualified practitioner regarding the treating physician or practitioner intent for the test. The date the service was performed should be stated in the patient’s record. Labs should also review and maintain all patient medical records supporting test as reasonable and necessary.
AltuMED PracticeFit enables Medical Billers with an inbuilt scrubber feature, reducing the chances of coding errors to 92%. Schedule a PracticeFit demo to know more:
Subscribe to Our Newsletter!
Subscribe to Our Newsletter!
Enter Your Email Address. We Promise We Won't Spam You
Relevant Articles
Categories
Follow Us